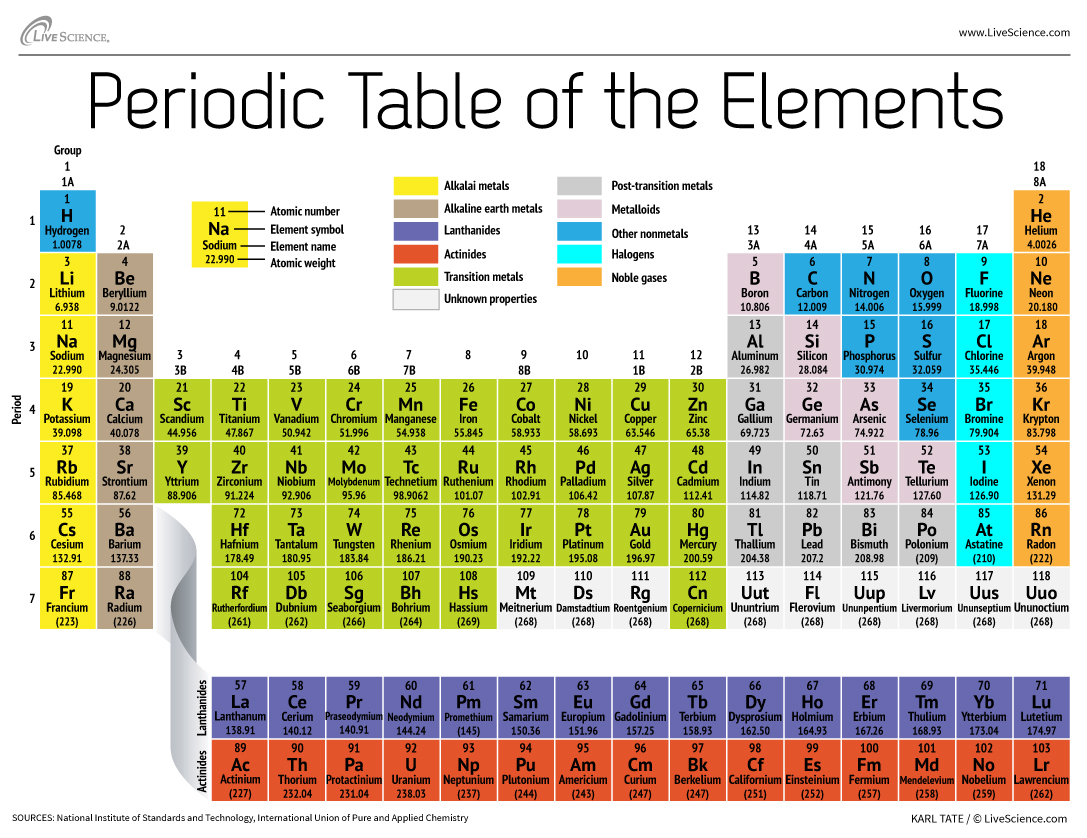
 After the Hindenburg blimp explosion, helium replaced hydrogen in blimps to prevent future explosions!Įach of these families has a unique set of characteristics based on the atomic structure of the atom. Noble gases are also used in medical products, providing stability instead of reactivity. Image by Alchemist-hp based on images by Jurii, via Wikimedia Commons, is licensed under Creative Commons Attribution 3.0 Unported license. Check out the different colors of the noble gases: Helium only has two electrons in the outer shell, but that is because it is such a small atom, with only two protons!ĭue to the low reactivity of these elements, they are used for purposes where reactivity could be damaging. Noble gases include helium, neon, argon, krypton, xenon, and radon. This stability causes the elements to have low reactivity. Group 18, or the noble gases, are elements that naturally have eight electrons in the outer shell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
